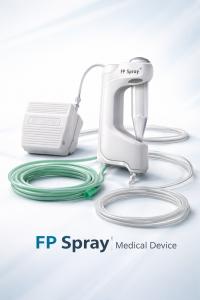
A pioneering Taiwan-based medical device startup, today announced its strategic expansion into the Southeast Asian market.
TAINAN CITY, TAIWAN, May 6, 2026 /EINPresswire.com/ — Endo Peace Biomedical Technology Co., Ltd., a pioneering Taiwan-based medical device startup, today announced its strategic expansion into the Southeast Asian market. The company will showcase its flagship Functional Powder Spray System at the SEA Healthcare & Pharma Show 2026, taking place in Kuala Lumpur, Malaysia, from May 20 to 22.
This milestone marks Endo Peace’s first international exhibition in the region, representing a pivotal step in bringing its innovative endoscopic solutions to global healthcare professionals.
Redefining Endoscopic Treatment: A Powder-Based Platform
The Functional Powder Spray System is a “Taiwan-first” innovation designed to overcome the limitations of traditional mechanical instruments and liquid-based formulations. By leveraging gas dynamics and fluid transport mechanisms, the system enables non-contact, rapid, and precise delivery of therapeutic powders directly to gastrointestinal lesions.
Key technological highlights include:
Open-Platform Architecture: Unlike closed systems, this platform is compatible with a diverse range of functional powders, including pharmaceuticals, medical device powders, traditional herbal extracts, and probiotics.
Preservation of Drug Integrity: The system delivers medications in their original state through simple physical preparation—such as grinding tablets—eliminating the need for complex reformulation.
Universal GI Application: Engineered for versatility, the system can be applied across the esophagus, stomach, small intestine, and colon.
Commitment to Quality and Regulatory Excellence
Endo Peace enters the Southeast Asian market with a robust foundation of compliance. The company has secured its medical device manufacturing license from the Taiwan Food and Drug Administration (TFDA) (No. MOHW-MD-008448) and is certified under ISO 13485:2016. To further its regional footprint, the company is actively pursuing medical device registrations in Malaysia and Thailand.
Leadership Insight
“To master modern hemostasis technology, one must embrace emerging technologies that transcend traditional boundaries,” said Xi-Zhang Lin, CEO of Endo Peace Biomedical and Honorary Professor at National Cheng Kung University. “Our platform does not just offer a product; it opens a new field of disease management through customizable, powder-based delivery.”
About
Endo Peace Biomedical is a Taiwan-based startup dedicated to the development of innovative medical devices that bridge the gap between clinical needs and engineering expertise. The company is focused on platforming endoscopic therapy to provide scalable, intuitive, and high-impact solutions for the global healthcare market.
Visit us at SEA Healthcare & Pharma Show 2026:
Dates: May 20–22, 2026
Booth: 6G09, Hall 6
Venue: Kuala Lumpur Convention Centre
For more information, please visit: https://www.endo-peacebio.com.tw/
Media Contact:
Mr. Xi-Zhang Lin / Mr. Po-Ming Yang
Endo Peace Biomedical Technology Co., Ltd.
endopeacebio@gmail.com
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()